What are LNPs?
First developed in 1990 as an alternative to carriers such as liposomes and polymeric nanoparticles, lipid nanoparticles (LNPs) have become the go to delivery vessel for nucleic acid based drugs among others. With several FDA approved LNP formulations already on the market and others in clinical trial, they permit the delivery of therapeutic nucleic acid payloads, including the extensively used mRNA based COVID-19 vaccines which saw them first emerge into general public consciousness. Prominent examples of FDA-approved LNP formulations include ALC-0315, SM-102 and Dlin-MC3-DMA with many more novel formulations in development today.
LNPs are typically spherical with a diameter of 60-100 nanometers and are comprised of five components[1]:
- Ionizable (cationic) lipid
- Helper phospholipid
- Polyethylene Glycol (PEG)-lipid
- Sterol (Cholesterol)
- Nucleic acid payload
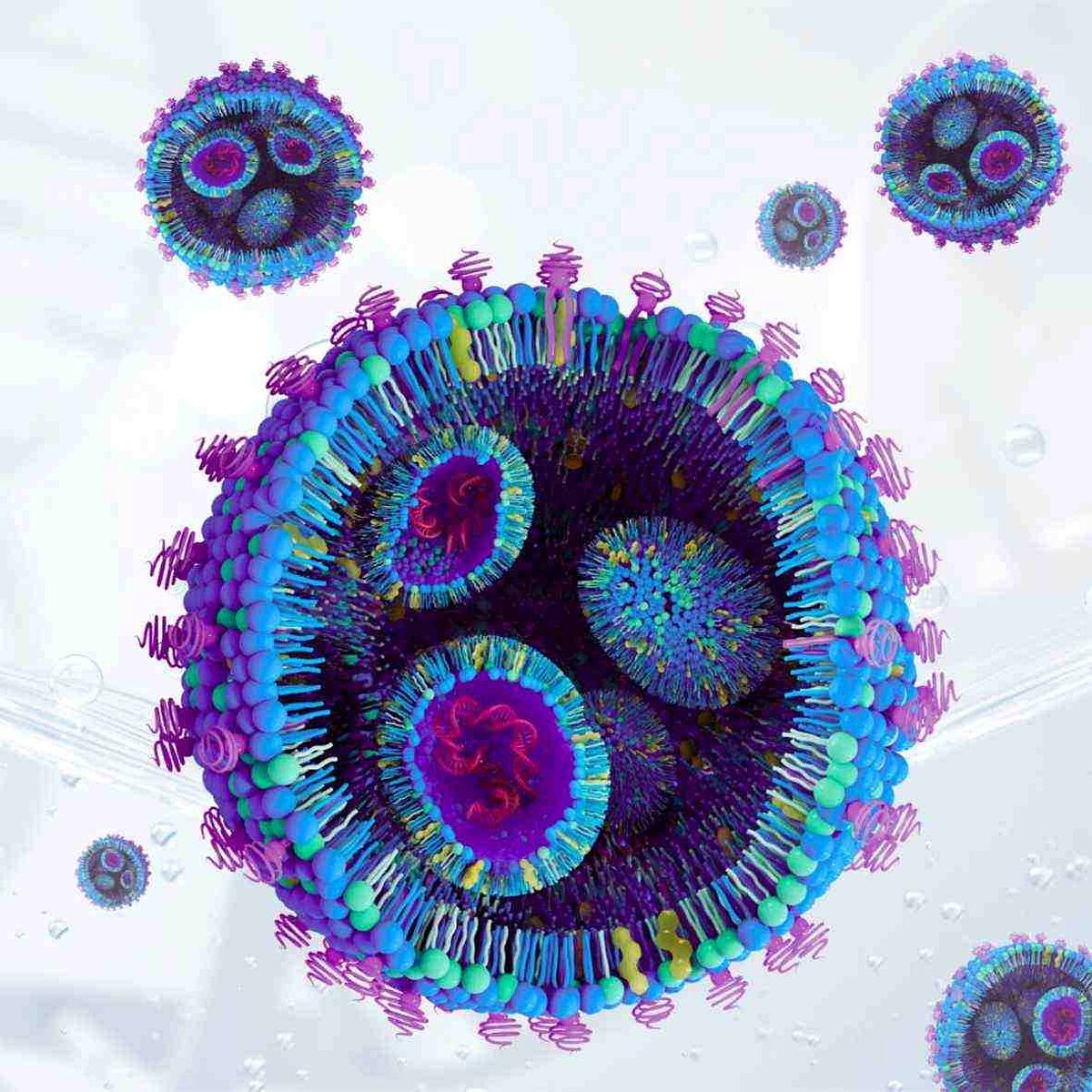
Lipid composition is the most critical part of LNP formulation, but each component plays a vital role in ensuring their fitness for purpose. Nucleic acid incorporation into the LNP is made possible via the overall positive charge of the LNP in acidic conditions. This promotes attractive electrostatic interactions with the negatively charged phosphate backbone of the nucleic acids enabling efficient nucleic acid encapsulation. When the LNPs are moved into a physiological pH of ~7.4 they remain of neutral charge promoting endosomal uptake and delivery of the nucleic acid payload into the cells. This neutral state at physiological pH means they have fewer interactions with blood cells therefore exhibiting lower toxicity and higher biocompatibility[2,3]. The additional phospholipids, cholesterol and PEG-lipids all assist with the stability of the final product, enabling long term storage and increased circulation time within the bloodstream.
Applications of LNPs – Vaccine, C> and beyond
Lipid Nanoparticles are sophisticated delivery vehicles, able to both protect fragile drugs and deliver them precisely to target tissues. Known for their functionality in the delivery of mRNA based vaccines, their capabilities extend far further. They are effective in delivering many types of nucleic acids, hydrophobic small molecule APIs, peptides, proteins and gene-editing systems[4] making them an extremely versatile drug delivery platform with great future application.
Recent RNA-LNP constructs have been used to treat cancers as adjuvants, neoantigen vaccines, CAR-T and epigenetic regulators as well as hereditary diseases as genetic editors. They are also used in the delivery of cancer vaccines and to deliver linear mRNA-based vaccines to infectious diseases such as malaria, HIV and influenza[5].
Of increasing interest is the use of LNPs as an alternative to viral vector delivery for CRISPR therapies. The majority of CRISPR-based therapeutics today are nucleic acid-based and have traditionally been delivered using viral vectors (often AAVs), which have shortcomings such as restricted payload capacity and patient immune responses to the AAV capsid. Delivering these therapies via LNP however offers several advantages including ease of manufacture (once composition is optimised), low immunogenicity and their transient nature.
A significant advantage of LNPs with regard to CRISPR therapy delivery is their lower immunogenicity profile than that of the AAVs traditionally used. Their lower immunogenicity makes it possible to deliver multiple doses of the therapy, something that just isn’t possible with an AAV delivery methodology. In addition, the transient nature of LNPs induces high CRISPR protein expression following delivery but reduces over time therefore lessening the likelihood of unintended off target edits to DNA at non-target sites in the genome[6].
The success in 2025 at the Children’s Hospital of Philadelphia, where a ground-breaking single-patient clinical trial for the world’s first-in-human personalised LNP delivered CRISPR therapy paves the way for rapid-response, personalised gene-editing therapeutics. Emerging interests include extending LNP-based CRISPR therapies beyond genetic conditions and into oncology and cardiovascular disease. Further work to streamline development and manufacturing will help lower costs and extend patient accessibility to these ground-breaking treatments.
Challenges
Despite their obvious flexibility and advantages as drug delivery vehicles, LNPs and their ongoing development must overcome several challenges. The greatest challenge is that of inherent liver tropism, which currently restricts their broader therapeutic applications. To achieve extra-hepatic targeting, researchers are engineering the composition of LNPs via the ratios and formulations of ionizable and helper lipids, cholesterol and other components. They are also investigating LNP surface modifications including the utilisation of targeting ligands including antibodies, peptides and aptamers to allow LNPs to target organs other than the liver[7].
Additional challenges for LNP applications include that availability of suitable lipid sources for scaled LNP production. With the focus on optimising LNP formulation to deliver payload to the specific tissue of interest it is essential to continue to screen multiple lipids of both natural and synthesised origin. Several organisations are working to provide a variety of tools to support this research including Cayman Chemicals and Abitec Larodan Research Grade Lipids. Products from both companies are available from LubioScience.
References
- Schober, G.B., Story, S. & Arya, D.P. A careful look at lipid nanoparticle characterization: analysis of benchmark formulations for encapsulation of RNA cargo size gradient. Sci Rep 14, 2403 (2024).
- Hou, X., Zaks, T., Langer, R. et al. Lipid nanoparticles for mRNA delivery. Nat Rev Mater 6, 1078–1094 (2021). https://doi.org/10.1038/s41578-021-00358-0
- Let’s talk about lipid nanoparticles. Nat Rev Mater 6, 99 (2021). https://doi.org/10.1038/s41578-021-00281-4
- https://www.pharmasalmanac.com/articles/expanding-lnp-applications-beyond-mrna-vaccines
- Vasileva O, Zaborova O, Shmykov B, Ivanov R, Reshetnikov V. Composition of lipid nanoparticles for targeted delivery: application to mRNA therapeutics. Front Pharmacol. 2024 Oct 23;15:1466337. doi: 10.3389/fphar.2024.1466337. PMID: 39508050; PMCID: PMC11537937.
- www.drugtargetreview.com/article/188056/advancing-gene-editing-the-role-of-lipid-nanoparticles-in-crispr-delivery/
- Truong HQ, Meng F. Unlocking the full therapeutic potential of lipid nanoparticles through extrahepatic delivery. Nano Research, 2025, 18(5): 94907422. https://doi.org/10.26599/NR.2025.94907422
Featured suppliers

Cayman Chemical
Cayman Chemical provides researchers with high-quality research tools, including lipids, biochemicals, assay kits, antibodies, proteins, and certified reference materials. Specializing in areas like cancer, neuroscience, metabolism, and drug discovery, Cayman supports scientific advancement through expert synthesis, assay development, and protein research.

Larodan Lipids
Larodan Lipids develops and manufactures a comprehensive range of high quality, research grade lipids and mass spectrometry standards, manufactured by both extraction and purification from natural sources as well as by chemical synthesis.
