Plasmid purification quality is often judged using basic spectrophotometric readouts, such as the A260/280 ratio, which mainly indicate protein contamination. While useful, this metric captures only a small portion of overall DNA quality. In reality, truly high‑quality plasmid DNA must be free from more than just proteins: it should also lack endotoxins, genomic DNA, RNA, and residual salts. These contaminants can easily evade routine checks yet still have a substantial impact on downstream experiments.
A broader and more practical definition of clean plasmid DNA therefore includes the elimination of any substance capable of disrupting enzymatic activity, compromising cell viability, or introducing unwanted variability into experimental workflows.
How impurities influence qPCR, NGS, and transfection workflows
Many downstream applications are acutely sensitive to impurities that may not be evident during standard quantification.
In mammalian cell transfection experiments, endotoxin contamination is a critical factor. Even trace levels can reduce cell health and impair DNA uptake, resulting in variable expression outcomes despite acceptable plasmid yields.
Similarly, qPCR and next‑generation sequencing workflows can be influenced by contaminants such as residual salts, chaotropic agents, RNA, or genomic DNA. These impurities may interfere with reaction kinetics or skew library preparation, often appearing as lower amplification efficiency, greater replicate‑to‑replicate variation, or reduced sequencing performance.
When plasmid DNA fails to meet the required purity thresholds, downstream inconsistency can mask the true behavior of reagents, constructs, or experimental conditions.
The operational cost of inadequate plasmid purity
Poor plasmid purity frequently leads to repeated transfection attempts, extended optimisation efforts, and unnecessary troubleshooting. These activities consume valuable reagents, time, and researcher effort that could be better spent advancing experimental objectives. In many cases, the root cause lies in upstream DNA quality rather than downstream protocols, but this link often becomes apparent only after multiple failed experiments.
Starting with high‑quality plasmid DNA is therefore a key contributor to laboratory efficiency and robust biological interpretation.
How ZymoPURE™ II supports consistency and reproducibility
Zymo Research’s ZymoPURE™ II plasmid purification kits are designed to directly address these challenges. By combining a silica‑based purification approach with an integrated endotoxin‑reduction workflow, the system effectively removes impurities that interfere with sensitive biological assays. The resulting plasmid DNA is well suited for demanding applications such as transfection, qPCR, and next‑generation sequencing.
One of the key strengths of the ZymoPURE™ II system is its reproducibility. The workflow delivers consistent plasmid purity and performance across users, batches, and laboratories, helping to minimise variability that can arise from manual handling or incomplete contaminant removal.
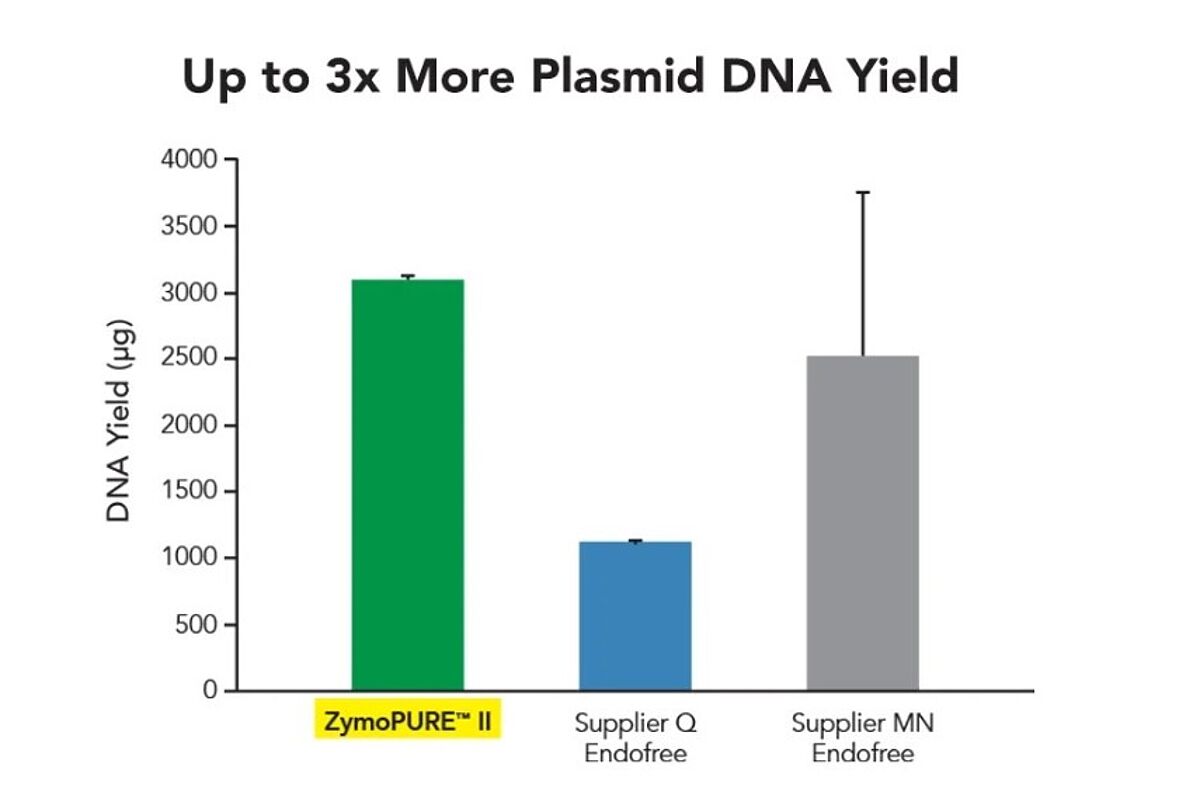
The link between plasmid purity and experimental outcomes
High‑quality plasmid DNA provides a reliable foundation for downstream experiments. When impurities are effectively removed, transfection outcomes are more uniform, expression levels become more predictable, and sequencing or qPCR workflows operate with greater consistency. This reliability allows researchers to interpret results with confidence and significantly reduces the need for repeated optimisation.
Clean input DNA supports clean, interpretable biological outcomes. By choosing purification methods that address all relevant contaminants, researchers can ensure that plasmid quality enhances, rather than limits, the performance of their experiments.
Curious? Try the market leader for free!
Zymo Research's kits save time, effort and money. Try their kits for free by filling out the form below.

Featured supplier

Zymo Research
Zymo Research is a leader in molecular biology, offering a comprehensive range of products for DNA, RNA, and epigenetics research. Established in California in 1994, the company is renowned for its high-quality nucleic acid purification technologies, including kits and reagents for DNA and RNA clean-up, isolation, and sequencing. Zymo is also a pioneer in epigenetics, with products for DNA methylation analysis, chromatin analysis, and NGS library preparation. Each product is designed to be simple to use, reliable, and available at competitive prices, making them ideal for both academic and biopharmaceutical research.
